 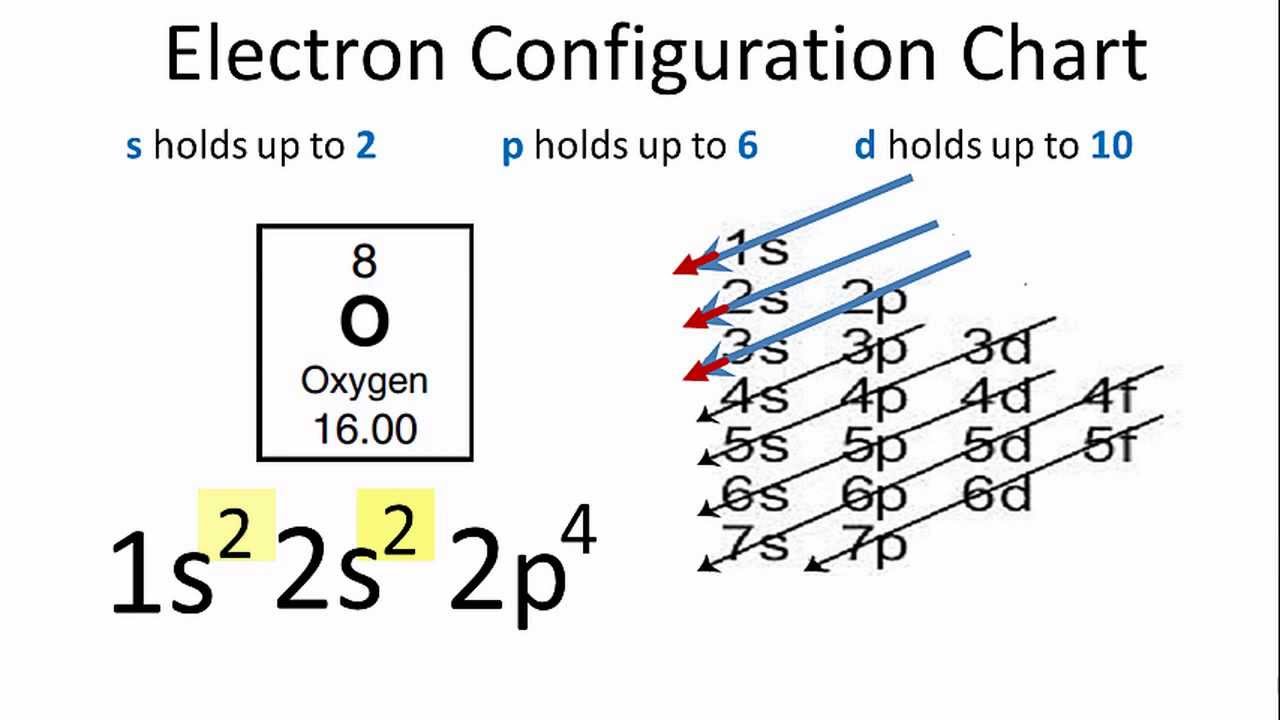
The aufbau principle tells you to fill in the order $\ce$ orbital when discussing the electronic configuration of charged d-block elements. In these cases, a completely full or half full d sub-level is more stable than. See for example this question to catch a small glimpse of the truth behind the simplifications. There are two main exceptions to electron configuration: chromium and copper. In all of these scenarios, the new redistribution of the electron is energetically favorable compared to the predicted configuration.Chromium’s standard electron configuration as given by the aufbau principle is actually more of an exception than you may think initially. You will see a similar situation where an #s# electron moves to a #d# sublevel with Molybdenum: 1) write out the entire electron configuration for chromium using sped notation. The short-hand (or condensed) electron configuration is (Ar). So, in this case, an electron moves to #3d# and is unpaired, therefore maximizing the spin state. Become a member to unlock this answer Create your account. The full electron configuration of chromium (Cr) is 1s2 2s2 2p6 3s2 3p6 4s1 3d5. Since two opposite spins result in a total spin of #0#, maximizing this tends to require as many electrons in of same spin in different orbitals as possible. Hund's Rule: It is energetically favorable to maximize the spin state in a sublevel.The #3d# orbital is slightly lower in energy, and minimizing repulsions in the #4s# orbital by moving one of the #4s# electrons to a close-lying #3d# orbital minimizes the ground-state energy of chromium.Ag has 2 more d electrons and 1 less s electron than Pd. Write MO electron configurations for F2, F2-, F2+. Write the full electron configuration for gold. The explanation on why chromium displays such a high turnout of reflected photon waves in general. This occurs again later in the periodic table with other elements and their electron configurations, such as copper, niobium. Write down the electron configuration of the Cu(I) ion. Chromium is a chemical element with the symbol Cr and atomic number 24. Sb3-Write the electron configuration for a beryllium atom. Ag has 1 more d electron and the same number of s electrons as Pd. Condensed electron configuration Write the condensed electron configuration for the following pairs: a. Ag has 2 more d electrons and the same number of s electrons as Pd. This happens in Chromium, as one #4s# electron moves to the #3d# sublevel. A comparison of the electron configurations of palladium (Pd) and silver (Ag) indicates that: A. Chromium exhibits +2, +3 and +6 oxidation states. 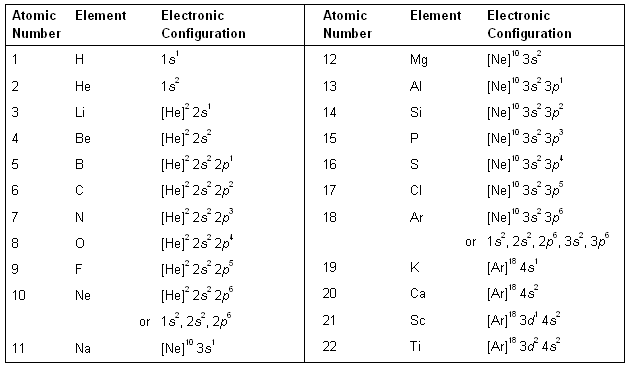
This actually allows electrons to move between the sublevels if it is energetically favorable. The electron configuration of chromium ion(Cr3+) is 1s2 2s2 2p6 3s2 3p6 3d3. Even though #4s# belongs to a higher main energy level than #3d#, the energy levels are similar. it appears that the most stable configuration for atoms of chromium, copper, niobium, molybdenum, ruthenium, rhodium, silver, platinum and gold involves only moving one electron into an s orbital. So why is this? It has to do with main the arrangement of sublevels in the electron cloud. However, the actual configuration of Chromium is: Therefore the expected electron configuration for Chromium will be 1s 2 2s 2. cypress run -config pageLoadTimeout100000,watchForFileChangesfalse. The words are used in the correct sentence according to their meaning in a. Looking at the periodic table, you would expect Chromium to have this configuration: The browser argument can be set to chrome, chromium, edge, electron, firefox. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
